- Inicio
- »
- Circuito de almacenamiento y distribución de agua para inyección (WFI)
Circuito de almacenamiento y distribución de agua para inyección
El circuito de almacenamiento y distribución de agua para inyección (WFI) de MOLECULAR es un sistema diseñado con gran precisión para el almacenamiento y suministro seguros y estériles de agua para inyección. Esta solución, que cumple con las buenas prácticas de fabricación (GMP), garantiza la pureza constante y la temperatura estable del agua para inyección en aplicaciones farmacéuticas críticas.
Características principales de nuestro circuito de almacenamiento y distribución de agua para fabricación de medicamentos (WFI)
Materiales de alta calidad
El sistema utiliza acero inoxidable 316L de grado sanitario, cuidadosamente seleccionado para evitar la lixiviación de iones de la pared de las tuberías y garantizar la integridad del agua para inyección (WFI) almacenada.
Control preciso de la temperatura
Se ha integrado una unidad de control de temperatura de un solo punto o de varios puntos para regular con precisión la temperatura del agua, lo que permite mantener un entorno estable y estéril.
Circulación óptima
El sistema cuenta con un circuito de distribución continua de agua para uso farmacéutico (WFI) con un caudal controlado por un variador de frecuencia (VFD), lo que inhibe eficazmente el crecimiento microbiano al evitar el estancamiento del agua.
Diseño de bajo consumo
Un diseño exclusivo de calefacción eléctrica y circulación está optimizado para mantener una temperatura de circulación estable y, al mismo tiempo, reducir al mínimo el consumo de energía.
Ventajas de nuestro circuito de almacenamiento y distribución de agua para inyección (WFI)
Mayor pureza y esterilidad: La combinación de un diseño higiénico, una circulación óptima y un control preciso de la temperatura garantiza que el agua para inyección (WFI) almacenada se mantenga pura y libre de contaminación microbiana, cumpliendo así con los estrictos requisitos normativos.
Cumplimiento y validación: Nuestro sistema está diseñado para cumplir con las normas cGMP, lo que facilita los procesos de validación. Los componentes de alta calidad y su diseño garantizan un rendimiento fiable a largo plazo.
Funcionamiento energéticamente eficiente: Los diseños optimizados de calefacción y circulación, que ahorran energía, permiten reducir los costos operativos en comparación con los sistemas tradicionales.
Menor mantenimiento: Al impedir el crecimiento microbiano y la formación de sarna, el sistema reduce al mínimo la necesidad de limpiezas y mantenimiento frecuentes, lo que garantiza un tiempo de funcionamiento constante.
Modelo de producto: Sistemas WFI
| No. | Modelo | Capacidad |
| 1 | MOLWFI-100L | 100 l/h |
| 2 | MOLWFI-200L | 200 l/h |
| 3 | MOLWFI-300L | 300 l/h |
| 4 | MOLWFI-500L | 500 l/h |
| 5 | MOLWFI-1000L | 1000 l/h |
| …… | ……. | …… |
| 6 | MOLWFI-10000L | 10 000 l/h |
Aplicaciones de los sistemas de almacenamiento y distribución de agua para inyección (WFI)
Este circuito de almacenamiento y distribución de agua para inyección (WFI) de gran fiabilidad es un componente esencial en cualquier instalación que requiera un suministro constante de agua de alta pureza.
Fabricación de productos farmacéuticos
Imprescindible para el almacenamiento y la distribución de agua para inyección (WFI) a las líneas de producción destinadas a la fabricación de productos inyectables y estériles.

Biotecnología y bioprocesos
Se utiliza para suministrar agua pura en la preparación de medios de cultivo celular y en otras etapas críticas de los bioprocesos.

Farmacias hospitalarias
Proporciona una fuente estéril de agua para inyección (WFI) para la elaboración de preparaciones estériles y otras aplicaciones farmacéuticas.

El principio de funcionamiento del circuito de almacenamiento y distribución de agua para inyección (WFI)
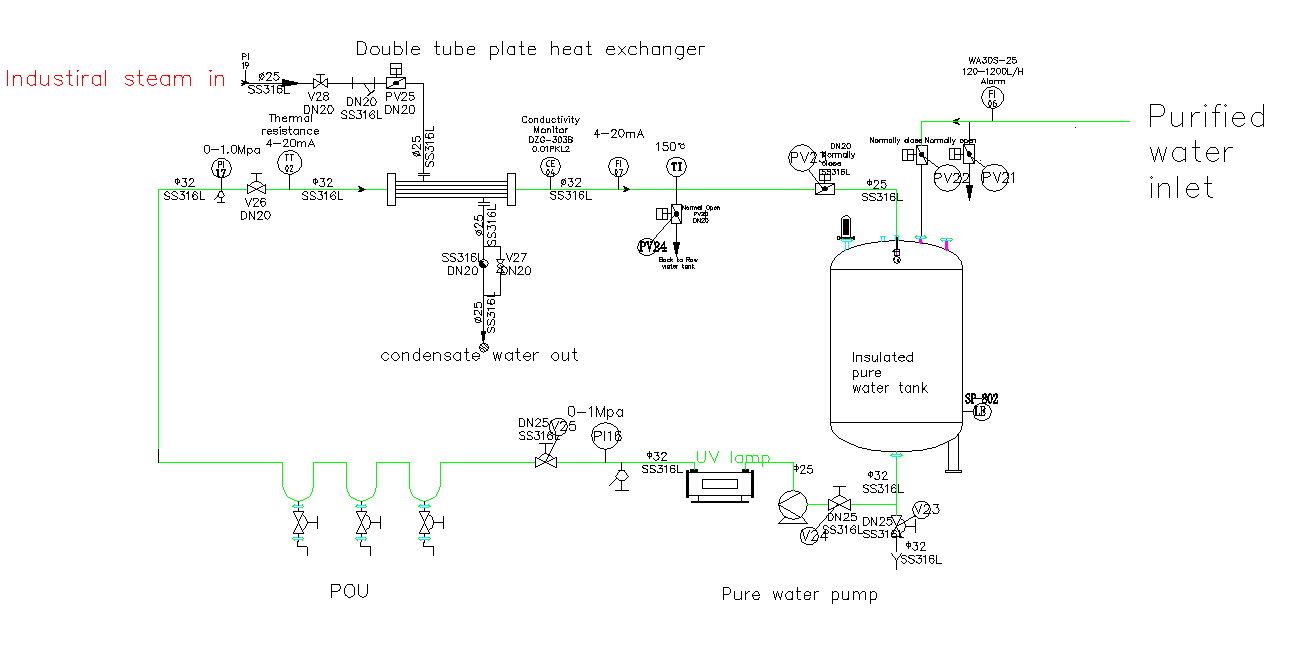
El circuito de almacenamiento y distribución de agua para inyección (WFI) es un sistema fundamental en la fabricación de productos farmacéuticos, diseñado para almacenar y suministrar de forma continua agua de alta pureza, al tiempo que evita el crecimiento microbiano.
El principio fundamental consiste en la circulación constante de agua para uso farmacéutico (WFI), que normalmente se mantiene a una temperatura superior a los 70 °C, por toda la instalación. Esta alta temperatura desinfecta eficazmente el sistema e inhibe el crecimiento microbiano. Una bomba de circulación mantiene el agua fluyendo a alta velocidad, creando un flujo turbulento que evita la formación de zonas de estancamiento donde podrían proliferar las bacterias.
El circuito está fabricado en acero inoxidable 316L de alta calidad, con superficies internas electropulidas para minimizar la rugosidad superficial y el riesgo de formación de biopelículas. Se supervisan continuamente parámetros clave como la temperatura, la conductividad y la presión para garantizar que la calidad del agua cumpla con las estrictas normas de la farmacopea antes de su uso en los procesos de fabricación de medicamentos.
Preguntas frecuentes
Un circuito de almacenamiento y distribución de agua para inyección (WFI) es fundamental para mantener la alta pureza, la temperatura y la esterilidad del agua para inyección una vez generada, garantizando así que cumpla con estrictos estándares de calidad antes de su uso en la producción.
Nuestro circuito de distribución de agua para inyección (WFI) utiliza un diseño de circulación continua con un caudal controlado y una temperatura elevada y estable (normalmente >80 °C) para evitar el estancamiento del agua e inhibir el crecimiento de bacterias y otros microorganismos.
El tanque de almacenamiento y todo el circuito de almacenamiento y distribución de agua para inyección (WFI) están fabricados en acero inoxidable 316L de grado sanitario, un material conocido por su resistencia a la corrosión y su acabado superficial liso, lo que minimiza el riesgo de formación de sedimentos.
Sí, nuestro sistema está equipado con una unidad de control de temperatura precisa, de un solo punto o de varios puntos, para mantener el agua para inyección (WFI) a una temperatura específica y estable, lo cual es fundamental tanto para la esterilidad como para los procesos posteriores.
Por supuesto. Todo el sistema está diseñado y validado para cumplir con las buenas prácticas de fabricación actuales (cGMP) y otras normativas farmacéuticas pertinentes, lo que garantiza su idoneidad y conformidad para entornos de fabricación farmacéutica.
Artículos relacionados
Las 13 principales empresas de tratamiento de agua a nivel mundial
En la fabricación industrial moderna, el desarrollo de infraestructuras y la recuperación de recursos, los sistemas de tratamiento de agua actúan como los “pulmones” de una fábrica. Ya sea que necesite agua ultrapura para la limpieza de obleas de semiconductores, sistemas de descarga líquida cero (ZLD) para cumplir con estrictas normas medioambientales o unidades de filtración de agua en contenedores para proyectos en el extranjero, elegir al proveedor adecuado es una decisión fundamental para su negocio. Esto […]
7 datos clave: ¿Qué es la destilación de efecto múltiple para el agua para inyección (WFI)?
En el exigente mundo de la fabricación farmacéutica, la calidad del agua es imprescindible. Entre las diversas tecnologías de purificación, la destilación de efectos múltiples se erige como el referente del sector para la producción de agua para inyección (WFI). Si desea comprender cómo esta tecnología garantiza la seguridad de los pacientes y la eficiencia operativa, esta guía completa le explicará todo lo que necesita […]
5 razones por las que vale la pena tener un descalcificador de agua: guía profesional
Antes de profundizar en sus ventajas, es importante entender qué hace realmente un ablandador de agua. El agua dura contiene altas concentraciones de calcio y magnesio. Aunque no es perjudicial para la salud al beberla, estos minerales son perjudiciales para las tuberías. Un ablandador de agua profesional utiliza un proceso denominado «intercambio iónico». Dentro del tanque, hay perlas de resina recubiertas de iones de sodio […]
Póngase en contacto con nosotros
No dejes que los problemas de calidad del agua se interpongan en tu camino. El generador de agua purificada MoerWater te ayudará a resolver todos tus problemas de calidad del agua, mejorar la eficiencia operativa y garantizar que tus productos o resultados experimentales alcancen el mejor nivel posible.

*Nuestro equipo responderá a tus consultas en un plazo de 24 horas.
*Su información se mantendrá en la más estricta confidencialidad.








