Storage and Distribution System for PW WFI
In demanding industries such as pharmaceuticals, biotechnology, medical devices and microelectronics, purified water (PW) is an indispensable key medium in the production process. Its quality is directly related to the safety, effectiveness and final quality of the product. Therefore, a purified water storage and distribution circulation system that is well designed, efficient and complies with strict regulations is essential.
This page will provide an in-depth look at the core elements of a PW storage and distribution circulation system, including its design principles, key components, validation requirements, and how to optimize the system to ensure consistent water quality and regulatory compliance, helping you avoid potential product contamination and compliance risks.
Why choose Molewater solutions?
As an expert in high purity water, Molewater is committed to providing excellent purified water storage and distribution circulation systems to ensure that your production process meets the most stringent industry standards. We are fully aware of the importance of water quality to your products, so our systems are meticulously designed, material selected, and operated.
Excellent tank design
Vertical stainless steel tanks are used, with an inner surface polishing roughness of Ra<0.6 μm, which effectively avoids biofilm formation and ensures pure water quality from the source.
Intelligent breathing valve system
Equipped with air breathing valve or electric heated air breathing valve, it can accurately adjust the air pressure balance, protect the structural integrity of the water tank, and effectively prevent external contamination.
Complete natural drainage capability
The tank and piping system have excellent natural drainage capability, which minimizes the risk of microbial growth and ensures system hygiene.
Dynamic water quality maintenance
The advanced circulation design is adopted to maintain the dynamic flow of water quality, completely eliminate stagnant water areas, and continuously stabilize the water quality.
Multiple disinfection guarantees
Provide multiple periodic disinfection solutions such as ozone, ultraviolet (UV) or pasteurization to ensure that the microbial load is always within the controllable range.
Accurate water quality monitoring
Integrate advanced online water quality monitoring system to grasp key parameters such as conductivity and TOC in real time to ensure that water quality meets regulatory requirements.
Get a customized solution
We provide tailor-made solutions based on your specific needs and industry regulations (e.g. cGMP, USP, etc.) and ensure that the system is fully compliant.

Purified water storage and distribution system applications
Pharmaceutical industry
Production of drugs such as oral solutions, tablets, capsules, equipment cleaning, preparation of buffer solutions and culture media, etc.

Biotechnology industry
Cell culture, fermentation, protein purification, diagnostic reagent production, etc.

Medical device industry
Used for cleaning, sterilization and production processes of medical devices, etc.

Laboratory
Used for pharmaceutical laboratory reagent preparation, instrument cleaning, etc.

Microelectronics industry
Used for cleaning and cooling during chip manufacturing (usually requires higher grade ultrapure water, but PW is the basis).

Photovoltaic Industry
Polysilicon purification, silicon wafer cleaning in PV, and wafer fabrication in semiconductors, cooling water circulation etc.

Certification
Take action now to optimize your purified water system!
MoerWater provides expert solutions for pure water storage and distribution systems, whether you’re starting a new project or upgrading an existing one.
What is a purified water storage and distribution circulation system?
The purified water storage and distribution circulation system is a closed-loop system designed to store and deliver purified water that has been pretreated and purified (usually through reverse osmosis, EDI and other technologies) to various points of use in the production site while maintaining its established quality standards.
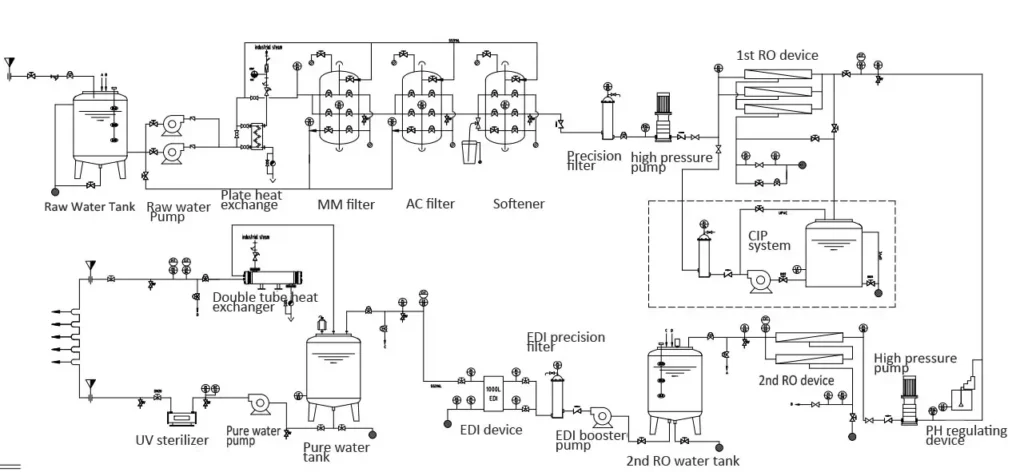
Core Components
- PW tank: used to store purified water, usually made of sanitary stainless steel, equipped with respirator, level gauge, spray ball, etc.
- Circulation pump: maintains continuous circulation of water in the system, ensures flow rate and pressure, prevents water retention and microbial growth. Usually adopts a redundant configuration of one for use and one for backup.
- Pipeline system: connects the storage tank and each use point to form a closed loop. High material requirements, usually sanitary 316L stainless steel.
- Valves and connectors: sanitary diaphragm valves, ball valves, etc., connected by clamps or orbital welding to ensure no leakage and dead corners.
- Online monitoring instruments: real-time monitoring of key water quality parameters such as conductivity, total organic carbon (TOC), temperature, flow and pressure.
- Ultraviolet (UV) sterilizer: usually installed on the return and/or supply pipelines to inhibit microbial growth.
- Heat exchanger (optional): used to adjust water temperature, especially when hot water disinfection or specific temperature requirements are required.

How it works?
Purified water enters the PW tank from the preparation system and is then transported to the entire distribution pipeline through a circulation pump. The water circulates continuously at high speed in the pipeline, passes through each use point, and then returns to the tank. The purpose of this continuous circulation is to:
- Maintain stable water quality: Prevent microbial growth and water quality deterioration caused by water retention.
- Ensure immediate supply: Ensure that each use point has a supply of water that meets the requirements at any time.
- Realize online monitoring: Real-time monitoring of water quality parameters, timely detection and handling of abnormalities.
The difference between PW and WFI:
Purified water (PW) and water for injection (WFI) differ significantly in quality requirements and system design. WFI has much higher requirements for microorganisms and endotoxins than PW, and is usually prepared by distillation or ultrafiltration. Its distribution system usually uses high temperature circulation (>80°C) or regular high temperature disinfection. To learn more about WFI systems, please click here.
FAQs About Purified Water System
Key strategies to prevent biofilm include: using sanitary grade 316L stainless steel materials with electropolished internal surfaces; designing pipelines with no dead legs and maintaining continuous slopes; ensuring sufficient circulation flow (turbulent flow); and performing regular, effective cleaning and disinfection (such as heat sterilization, chemical disinfection, or UV sterilization). MoerWater's storage tanks feature an internal surface roughness Ra < 0.6 μm and excellent natural drainage capabilities, effectively inhibiting biofilm formation.
Performance Qualification (PQ) for a PW system typically involves three phases. Phase I and Phase II each last 2-4 weeks, during which intensive sampling and testing are conducted. Phase III is the routine operation phase, with monitoring frequency determined by risk assessment. The entire validation process can last several months. Moore Water provides comprehensive validation support services.
Factors to consider include the supplier's industry experience, technical strength, understanding of compliance, customized design capabilities, equipment quality, automation level, after-sales service, and successful case studies. Moore Water demonstrates significant advantages in all these aspects.
Dead legs are areas where water flow stagnates, easily leading to microbial proliferation and biofilm formation, thereby contaminating the purified water. This can cause the water quality to fail to meet standards, severely impacting product quality and production compliance. Therefore, dead legs must be strictly controlled during design. Moore Water's system designs strictly adhere to the principle of no dead legs.
The frequency of sanitization for a PW system depends on several factors, including the system's design, operational parameters, raw water quality, and specific water quality monitoring results. Typically, periodic sanitization (e.g., weekly, bi-weekly, or monthly) is performed using heat (hot water or steam) or chemical agents (like ozone or peracetic acid) to control microbial growth. A robust monitoring program that includes total viable count (TVC) and conductivity measurements is crucial for determining the optimal sanitization schedule and ensuring compliance.
Upgrading a PW system to produce WFI quality water is complex and often requires significant modifications. While both systems use similar initial purification steps (like RO and deionization), WFI requires additional purification stages (e.g., multi-effect distillation, vapor compression distillation, or ultrafiltration with very tight membranes) and much stricter control over microbial and endotoxin levels, often necessitating hot storage and distribution loops. It's usually more cost-effective and compliant to design a dedicated WFI system rather than attempting a full conversion of a standard PW system.
Get in touch with us
Don’t let water quality issues stand in your way. MoerWater Purified Water Generator will help you solve all your water quality problems, improve operational efficiency, and ensure your product or experimental results are at the best level.

*Our team will answer your inquiries within 24 hours.
*Your information will be kept strictly confidential.




