- Home
- »
- WFI Storage And Distribution Loop
WFI Storage and Distribution Loop
The MOLECULAR WFI Storage and Distribution Loop is a meticulously engineered system designed for the safe and sterile storage and delivery of Water for Injection. This GMP-compliant solution ensures the continuous purity and stable temperature of WFI for critical pharmaceutical applications.
Key Features of Our WFI Storage and Distribution Loop
High-Quality Materials
The system utilizes sanitary-grade 316L stainless steel, carefully selected to prevent Ion Leaching from the Pipe Wall and ensure the integrity of the stored WFI.
Precise Temperature Control
A single-point or multiple-point temperature control unit is integrated to precisely regulate the water temperature, maintaining a stable and sterile environment.
Optimal Circulation
The system features a continuous WFI distribution loop with a VFD (Variable Frequency Drive) controlled flow rate, which effectively inhibits microbial growth by preventing water stagnation.
Energy-Saving Design
A unique electric heating and circulation design is optimized to maintain stable circulation temperature while minimizing energy consumption.
Advantages of Our WFI Storage and Distribution Loop
Enhanced Purity and Sterility: The combination of a sanitary design, optimal circulation, and precise temperature control guarantees that the stored WFI remains pure and free from microbial contamination, meeting stringent regulatory requirements.
Compliance and Validation: Our system is designed to meet cGMP standards, making validation processes straightforward. The high-quality components and design ensure long-term, reliable performance.
Energy-Efficient Operation: The optimized energy-saving heating and circulation designs result in lower operational costs compared to traditional systems.
Reduced Maintenance: By preventing microbial growth and rouge formation, the system minimizes the need for frequent cleaning and maintenance, ensuring consistent uptime.
Product Model-WFI Systems
| No. | Model | Capacity |
| 1 | MOLWFI-100L | 100L/H |
| 2 | MOLWFI-200L | 200L/H |
| 3 | MOLWFI-300L | 300L/H |
| 4 | MOLWFI-500L | 500L/H |
| 5 | MOLWFI-1000L | 1000L/H |
| …… | ……. | …… |
| 6 | MOLWFI-10000L | 10000L/H |
Applications for WFI Storage and Distribution Systems
This reliable WFI storage and distribution loop is a crucial component in any facility requiring a constant supply of high-purity water.
Pharmaceutical Manufacturing
Essential for storing and distributing WFI to production lines for injection and sterile product manufacturing.

Biotechnology & Bioprocess
Used to supply pure water for cell culture media preparation and other critical bioprocessing steps.

Hospital Pharmacies
Providing a sterile source of WFI for compounding sterile preparations and other pharmacy applications.

The Working Principle of WFI Storage and Distribution Loop
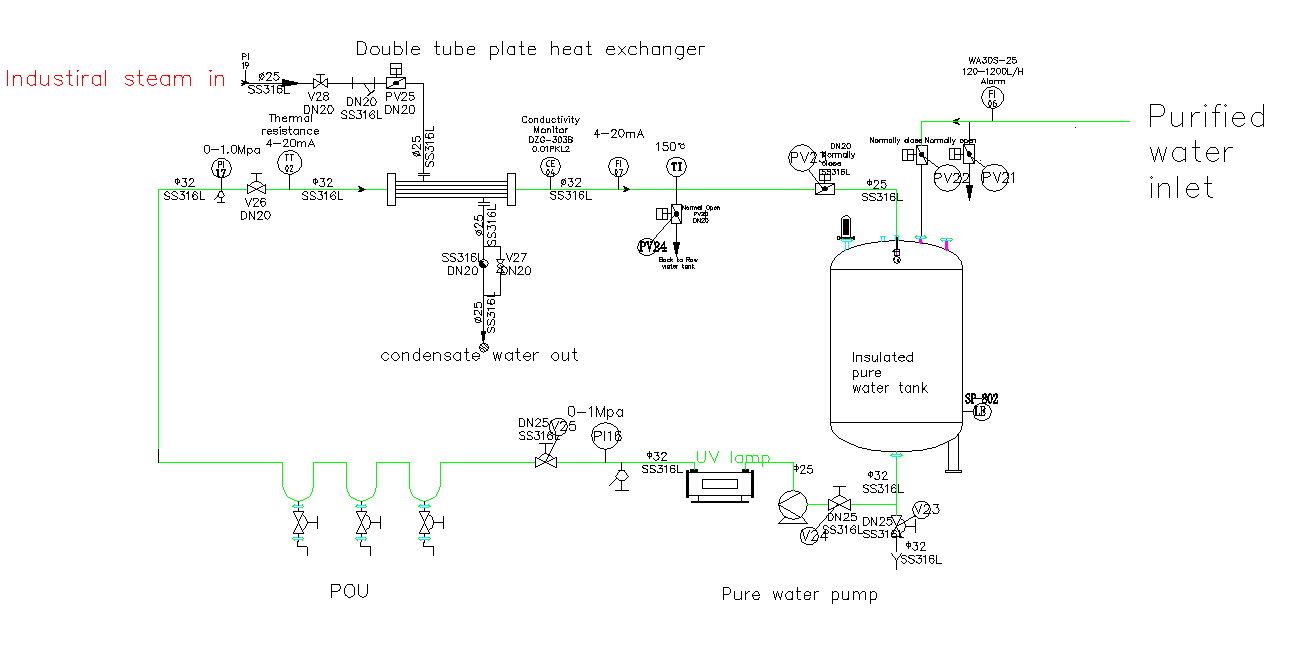
The WFI (Water for Injection) storage and distribution loop is a critical system in pharmaceutical manufacturing designed to continuously store and deliver high-purity water while preventing microbial growth.
The core principle involves constant circulation of WFI, typically held at a temperature above 70°C, throughout the entire facility. This high temperature effectively sanitizes the system and inhibits microbial growth. A circulation pump keeps the water flowing at a high velocity, creating a turbulent flow that prevents stagnant areas where bacteria could thrive.
The loop is constructed from high-quality 316L stainless steel with electropolished inner surfaces to minimize surface roughness and potential for biofilm formation. Key parameters like temperature, conductivity, and pressure are continuously monitored to ensure the water’s quality meets stringent pharmacopoeia standards before it is used in drug manufacturing processes.
Frequently Asked Questions
A WFI storage and distribution loop is vital for maintaining the high purity, temperature, and sterility of Water for Injection after it is generated, ensuring it meets strict quality standards before use in production.
Our WFI distribution loop uses a continuous circulation design with a controlled flow rate and stable high temperature (typically >80°C) to prevent water stagnation and inhibit the growth of bacteria and other microorganisms.
The storage tank and the entire WFI storage and distribution loop are constructed from sanitary-grade 316L stainless steel, a material known for its corrosion resistance and smooth surface finish, which minimizes the risk of rouge formation.
Yes, our system is equipped with a precise single-point or multiple-point temperature control unit to maintain the WFI at a specified, stable temperature, which is critical for both sterility and downstream processes.
Absolutely. The entire system is designed and validated to meet cGMP and other relevant pharmaceutical regulations, ensuring its suitability and compliance for pharmaceutical manufacturing environments.
Related Articles
7 Key Facts: What is Multi Effect Distillation for WFI?
In the stringent world of pharmaceutical manufacturing, the quality of water is non-negotiable. Among various purification technologies, multi effect distillation stands as the industry benchmark for producing Water for Injection (WFI). If you are looking to understand how this technology ensures patient safety and operational efficiency, this comprehensive guide will break down everything you need […]
5 Reasons a Water Softener Is Worth It: Pro Guide
Before diving into the value, it is important to understand what a water softener actually does. Hard water contains high concentrations of calcium and magnesium. While not harmful to drink, these minerals are destructive to plumbing. A professional water softener uses a process called ion exchange. Inside the tank, resin beads coated with sodium ions […]
Bottle Water Plant: Shocking 7 Startup Tips You Must Know
Introduction: Navigating the Bottle Water Plant Landscape Starting a bottle water plant is an endeavor filled with massive potential—and hidden pitfalls. In the highly competitive global bottled water market, success is determined not just by the quality of your source water, but by the efficiency and compliance of your operation. Many startups focus solely on […]
Get in touch with us
Don’t let water quality issues stand in your way. MoerWater Purified Water Generator will help you solve all your water quality problems, improve operational efficiency, and ensure your product or experimental results are at the best level.

*Our team will answer your inquiries within 24 hours.
*Your information will be kept strictly confidential.








