Agua para HPLC: el elemento vital invisible de la cromatografía
El agua para HPLC es, en esencia, el tipo de agua de laboratorio de mayor calidad, y suele clasificarse como Agua de tipo I. Debe cumplir tres requisitos imprescindibles:
1. Resistividad: la medida de la pureza iónica
Estándar: 18,2 MΩ·cm a 25 °C.
Importancia: Este valor representa la resistencia máxima teórica que puede alcanzar el agua pura, lo que confirma que las especies iónicas (sales, minerales) se han eliminado hasta el nivel más bajo posible. Esto garantiza que la pureza iónica no afectará a las separaciones por HPLC de fase inversa sensibles al pH.
2. Carbono orgánico total (COT): el archienemigo de la cromatografía
Estándar: Debe ser ultrabajo, normalmente ≤5 ppb (partes por mil millones).
Repercusión: Las moléculas orgánicas son la principal fuente de interferencia. En la elución en gradiente, los compuestos orgánicos en trazas presentes en el agua pueden concentrarse en la columna y luego eluirse más tarde, lo que provoca “picos fantasma” que comprometen la precisión del análisis cuantitativo. Por lo tanto, controlar el TOC es más importante que lograr una alta resistividad.
3. Absorbancia UV: la prueba definitiva de pureza
Requisito: El agua debe presentar un nivel mínimo Absorción de rayos UV en las longitudes de onda clave utilizadas en los análisis de HPLC y LC-MS (por ejemplo, 214 nm y 254 nm).
Validación: Una lectura de baja absorbancia confirma que el agua no contiene dobles enlaces conjugados ni compuestos aromáticos, lo que demuestra que realmente no produce interferencias. Esta es la comprobación práctica que distingue el verdadero Agua de calidad para HPLC del agua común de tipo I.

Los contaminantes que arruinan tus cromatogramas
Comprender el origen y los efectos de los contaminantes es fundamental para lograr agua de laboratorio gestión, especialmente en técnicas analíticas delicadas.
1. Impurezas orgánicas e inestabilidad de la línea de base
Los residuos orgánicos, la lixiviación de las tuberías, la exposición a los COV del aire ambiente o incluso las propias resinas de pulido pueden provocar una desviación de la línea de base o picos durante un análisis. En el análisis cuantitativo, estas fluctuaciones comprometen el límite de detección e introducen un error inaceptable. La eliminación de estas sustancias orgánicas es fundamental para lograr una línea de base plana y estable.
2. Partículas en suspensión y salud de la columna vertebral
Las partículas microscópicas, aunque a menudo no son de naturaleza química, suponen una amenaza física. Estos pequeños fragmentos pueden obstruir las delicadas válvulas de inyección del instrumento y los filtros sinterizados situados en la parte superior de la columna cromatográfica. El resultado es un aumento de contrapresión, formas de pico distorsionadas y una reducción drástica de la vida útil de la columna, lo que conlleva importantes costos de reemplazo y tiempo de inactividad. Por lo tanto, todos Agua para HPLC debe pasar por un proceso validado 0.22 µm filtro terminal.
3. Subproductos microbianos y endotoxinas
Aunque suele ser menos preocupante que en la producción farmacéutica, las bacterias pueden proliferar en el agua estancada agua de laboratorio. A medida que mueren y se descomponen, sus subproductos —que suelen ser orgánicos— se convierten en fuentes de COT, contaminando continuamente el flujo de agua y pudiendo formar biofilm dentro de las líneas de distribución.
Métodos avanzados de producción de agua de tipo I para HPLC
Para alcanzar y mantener el estándar de 18,2 MΩ·cm se requiere un sistema de purificación robusto y de varias etapas.
1. La Fundación Purificación
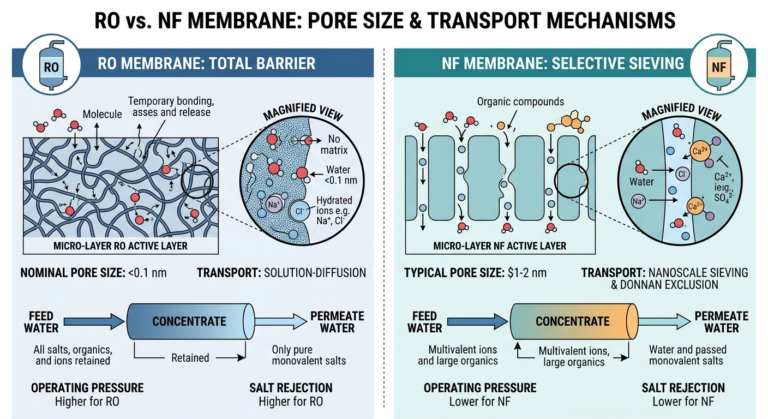
El proceso comienza con pretratamiento (eliminación del cloro y la dureza) seguida de Ósmosis inversa (RO) para eliminar el 95-99 % de los contaminantes. A continuación, el agua entra en el etapa de desionización (DI), a menudo mediante una combinación de resinas de intercambio iónico o electrodeionización (EDI), lo que eleva la resistencia hasta el máximo teórico.
2. Pulido de terminales: la clave para un bajo TOC
La etapa final es la que eleva el agua hasta Agua de calidad para HPLC eliminando los restos de sustancias orgánicas restantes:
Oxidación por rayos UV (185 nm): Esta potente luz ultravioleta de longitud de onda corta es el elemento clave para la eliminación del TOC. Tiene la energía suficiente para descomponer las moléculas orgánicas grandes residuales en pequeñas especies ionizadas (como el ácido carbónico).
Paquete de pulido de terminales: Specialized, resinas de intercambio iónico de grado nuclear luego captura estos fragmentos orgánicos recién ionizados, “purificando” eficazmente el agua hasta un nivel de TOC inferior a 5 ppb. Esta combinación de oxidación UV y resina nueva es esencial para cualquier sistema producción de agua para HPLC.
Ultrafiltración (opcional): En aplicaciones altamente sensibles, como la LC-MS (cromatografía líquida-espectrometría de masas), se puede añadir un ultrafiltro (UF) para eliminar macromoléculas, nucleasas y trazas de endotoxinas.

Control de calidad y mantenimiento para garantizar la pureza del agua en HPLC
Para obtener resultados analíticos fiables es necesario contar con Patrones de pureza del agua para HPLC. Un mantenimiento adecuado es la única forma de garantizar esta consistencia.
1. Validación: el protocolo de ensayo en blanco
Cualquier nueva fuente de agua o sistema de purificación que haya sido revisado recientemente debe verificarse directamente en el equipo de HPLC. El protocolo de ejecución en blanco consiste en utilizar agua purificada como fase móvil. El cromatograma debe mostrar un línea de base plana sin picos fantasma significativos en las longitudes de onda de detección operativas. Si el agua en sí misma introduce ruido, el análisis no es válido.
2. Mejores prácticas de mantenimiento
Sustitución oportuna: La vida útil del cartucho de pulido final determina directamente la Agua de calidad para HPLC. En el momento en que el monitor de TOC o la lectura de resistividad indique cualquiera Si se produce una fluctuación, hay que cambiar el cartucho.
Circulación continua: De alta pureza agua de laboratorio debe guardarse en circulación continua para evitar el estancamiento, que provoca rápidamente el crecimiento microbiano y la lixiviación de sustancias orgánicas de las tuberías.
Protección atmosférica: El grifo dispensador debe estar protegido por un filtro de ventilación para evitar que el CO₂ presente en el aire se disuelva en el agua (lo que reduciría la resistividad) y para impedir que los COV de la atmósfera del laboratorio entren en contacto con el agua.
Conclusión: La calidad del agua es la clave del éxito de la separación
Alcanzar los 18,2 MΩ·cm es solo el punto de partida para Agua para HPLC. La verdadera pureza cromatográfica requiere un sistema que esté diseñado, mantenido y validado para eliminar la amenaza silenciosa de los contaminantes orgánicos. Al invertir en un sistema específico Agua de tipo I Con un sistema de purificación que utilice oxidación UV y un mantenimiento adecuado del sistema, se asegurará de que el agua respalde —en lugar de comprometer— los resultados analíticos críticos.
¿Desea actualizar el sistema de agua de tipo I de su laboratorio para cumplir con los estándares críticos de HPLC/LC-MS? Póngase en contacto hoy mismo con nuestros expertos en tratamiento de agua para obtener un plan personalizado de asesoramiento y validación del sistema.