Why Your Dialysis Water Machine is Your First Line of Defense
The quality of your dialysis water directly impacts patient outcomes. Contaminants that are harmless in drinking water become deadly when bypassing the body’s natural defenses.
-
Bacteria & Endotoxins: Can cause pyrogenic reactions and septic shock.
-
Chemical Contaminants (Chloramine, Fluoride, Metals): Lead to long-term health issues like hemolytic anemia or neurodegenerative conditions.
-
Electrolytes & Minerals: Cause metabolic imbalances and cardiovascular instability during treatment.
Your dialysis water system is the critical barrier against these risks.

The Gold Standard: Demystifying AAMI/ISO 23500 for Dialysis Water
Any dialysis water machine must be designed, validated, and monitored to meet the rigorous AAMI/ISO 23500 series of standards. These define the allowable limits for chemical contaminants and microbial levels.
Key Parameters Include:
-
Action Levels for Bacteria: < 100 CFU/mL (Ultrapure Dialysate: < 0.1 CFU/mL)
-
Endotoxin Limits: < 0.25 EU/mL (Ultrapure Dialysate: < 0.03 EU/mL)
-
Chemical Contaminants: Strict limits for over 70 substances, including chloramines, heavy metals, and nitrates.
Anatomy of a Compliant Dialysis Water Purification System
A reliable system is a multi-barrier defense, with each component playing a vital role.
1. Pretreatment: The Essential Foundation
-
Carbon Filtration: Critically removes chloramines and chlorine, which are toxic to dialysis patients and can pass through reverse osmosis membranes.
-
Water Softening: Prevents scale buildup in the RO system, ensuring consistent performance and membrane longevity.
2. L'épuration primaire : Le cœur du système
-
Osmose inverse (RO) : The non-negotiable core technology. A high-rejection RO membrane removes over 99% of ionic, organic, and microbial contaminants, producing the “AAMI Quality” water standard for dialysis.
3. Distribution & Ultrapure Dialysate Generation
-
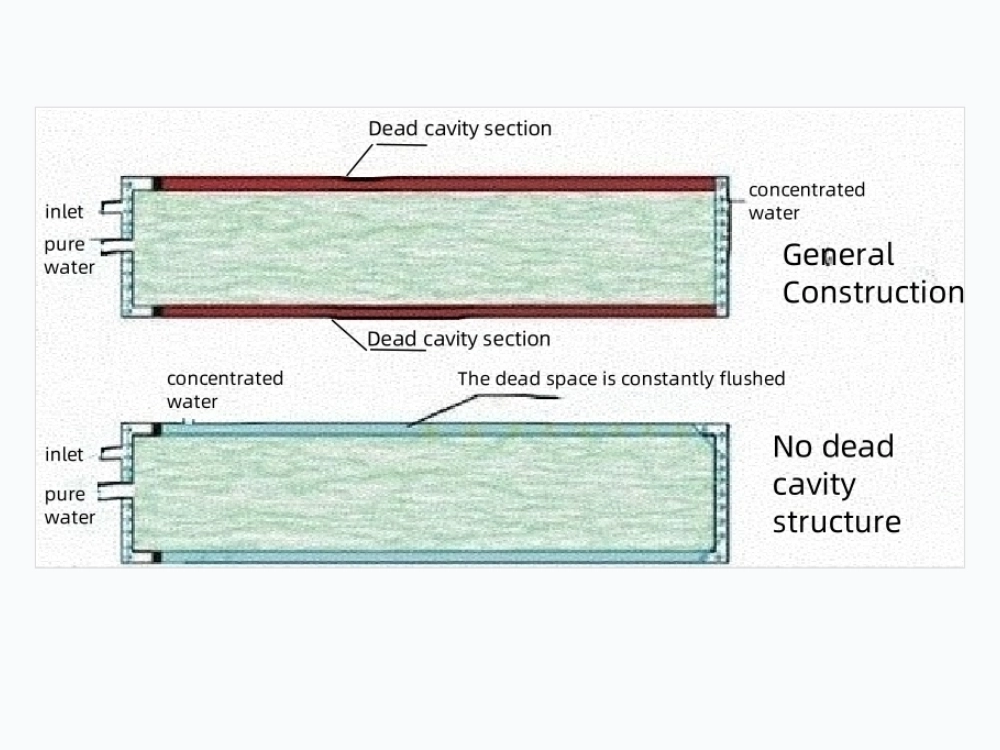
Sanitary Distribution Loop: Designed with 316L stainless steel or continuous PVC piping to prevent bacterial regrowth, featuring continuous recirculation.
-
Ultrafiltration (UF) Modules: Placed at the point-of-use, these filters are required to produce Ultrapure Dialysate, which is associated with reduced inflammation and better patient outcomes.
The 5 Critical Steps for Dialysis Water Machine Validation & Monitoring
Purchasing the equipment is only the beginning. Ongoing compliance is proven through rigorous protocols.
-
Initial Validation (IQ/OQ/PQ): Confirms the system is installed correctly and consistently produces AAMI-quality water.
-
Routine Microbial Monitoring: Weekly testing of bacteria and endotoxin levels at multiple points in the distribution loop.
-
Chemical Testing: Periodic (at least annual) full chemical analysis to verify RO membrane integrity.
-
Disinfection & Maintenance: Regular, documented chemical or hot water disinfection of the entire loop.
-
Comprehensive Documentation: Every action, test result, and maintenance procedure must be recorded to demonstrate compliance during audits.
L'avis d'un expert : The most common point of failure is not the RO itself, but inadequate pretreatment (especially carbon bed exhaustion) ou poor distribution loop sanitization, leading to biofilm formation and endotoxin spikes.
L'eau taupe Advantage: Your Partner in Dialysis Patient Safety
Au [Your Brand Name], we understand that your dialysis water machine is a life-sustaining device. We go beyond selling equipment to become your compliance partner.
-
AAMI-Centric Design: Notre dialysis water purification systems are engineered from the ground up to meet and exceed AAMI/ISO 23500 standards.
-
Validation-First Approach: We provide a complete validation package, including protocols and support for IQ, OQ, and PQ, making your accreditation process seamless.
-
Proactive Support & Monitoring: Our service plans include reminders for critical maintenance, like carbon bed replacement, and access to technical experts.
Are You Confident in Your Current Water Quality?
-
Struggling with persistent bacteria or endotoxin alerts?
-
Facing an upcoming state or accreditation audit?
-
Unsure if your current system can achieve Ultrapure Dialysate standards?

Take the First Step Towards Unshakeable Compliance and Patient Safety.
Click here to schedule a complimentary, no-obligation consultation with our water safety specialists to review your current system.