Introduction: When a Single Water Impurity Can Skew a Million-Dollar Experiment or Compromise a Diagnosis…
In the world of clinical diagnostics and life sciences research, water is not just a solvent—it’s the most critical reagent. The purity of water produced by your medical water purification system directly influences the accuracy of ELISA tests, the reliability of PCR results, and the safety of patient samples.
This definitive guide reveals how a state-of-the-art medical water system protects your laboratory’s most valuable assets: its data, its reputation, and the patients it serves.
The Unseen Cost of Inconsistent Medical Water Quality
Many labs operate on the brink of data integrity issues without realizing the source:
-
Failed Experiments & Wasted Reagents: Bacterial endotoxins or organic contaminants can inhibit cell cultures and enzymatic reactions, voiding weeks of research.
-
Inaccurate Diagnostic Results: Ionic interference can lead to false positives or negatives in critical clinical assays, directly impacting patient care.
-
Instrument Damage & Downtime: Scale buildup and particulates can clog sensitive instrumentation like HPLC and mass spectrometers, leading to costly repairs and halted workflows.
-
Regulatory Compliance Failures: Labs operating under CLIA ’88 ou CAP accreditation must demonstrate water quality meets strict standards like USP <643> et PE monographs.

The 3 Types of Laboratory Water Every Facility Needs
A modern medical water purification machine must be capable of producing the correct grade of water for its specific application.
-
Type III (RO Pure Water): For feed water to clinical analyzers, glassware washers, and environmental chambers.
-
Type II (eau pure) : For buffer preparation, media preparation, and general laboratory use.
-
Type I (eau ultra-pure) : For critical applications like HPLC, GC-MS, ICP-MS, and molecular biology.

Anatomy of a Reliable Medical Water Purification System
A compliant system is a multi-stage purification train, each stage eliminating a specific class of contaminants.
1. Pretreatment: The Foundation of Consistency
-
Carbon Filtration & Softening: Protects the downstream Reverse Osmosis (RO) membrane from chlorine and hardness, ensuring its longevity and efficiency.
2. L'épuration primaire : Le cœur du système
-
Osmose inverse (RO) : Removes 99% of ions, organics, bacteria, and pyrogens, producing consistent Type III/II water.
-
Électrodéionisation (EDI) : A chemical-free polishing technology that provides a continuous supply of high-purity water.
3. Final Polishing for Type I Ultrapure Water
-
Recirculating Ultrapure Loop: Features a UV lamp for TOC reduction and bacterial control, and a final ultrafiltration cartridge to remove any residual endotoxins and nucleases.
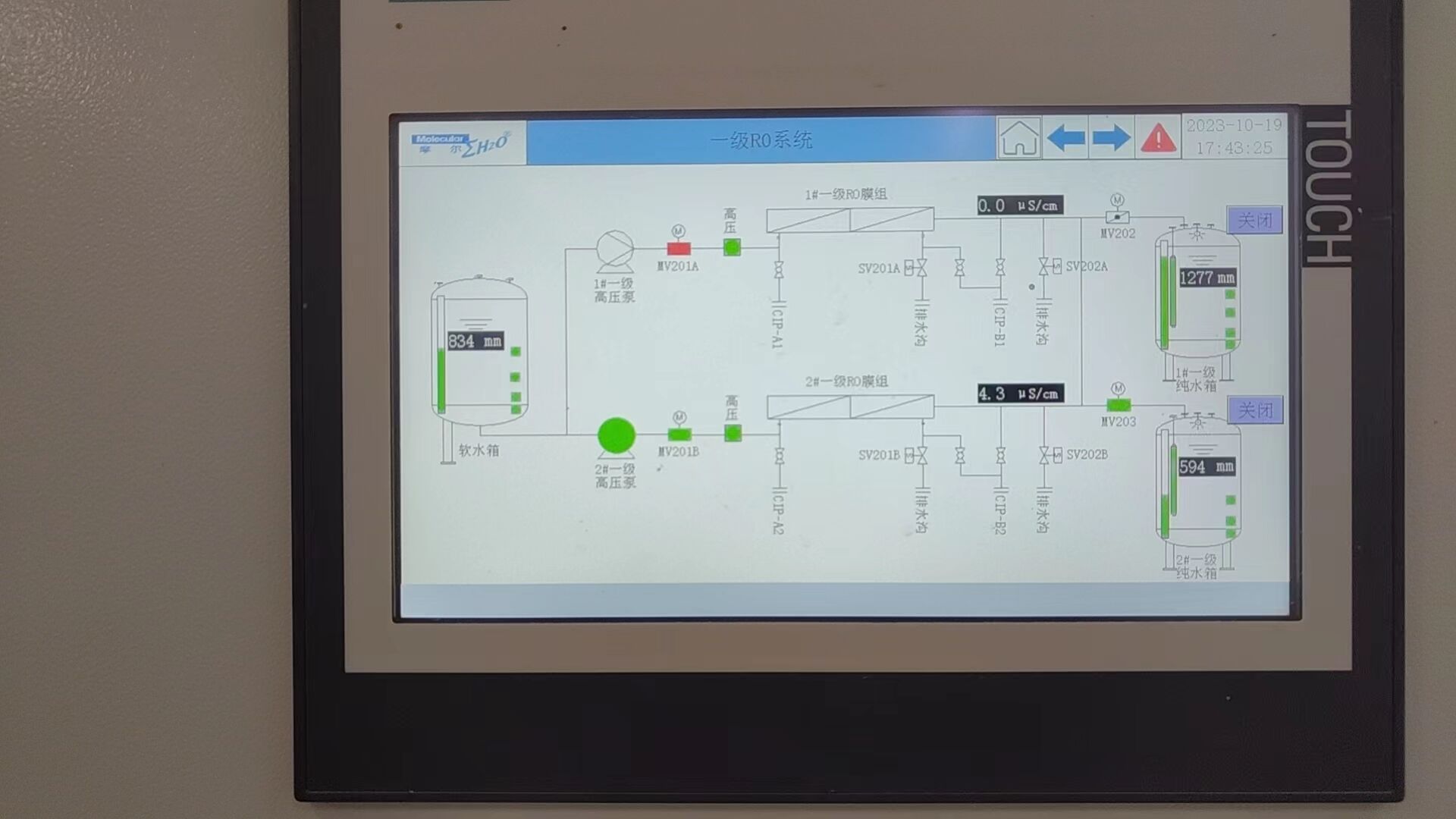
The 5 Non-Negotiable Steps for Validating Your Medical Water System
Installation is just the beginning. Ongoing compliance requires a rigorous, documented protocol.
-
Initial Qualification (IQ/OQ/PQ): Verifies proper installation and confirms the system consistently produces water that meets the required specifications (Type I, II, III).
-
Real-Time Purity Monitoring: Continuous tracking of Resistivity (18.2 MΩ.cm) and TOC (ppb) levels.
-
Routine Microbial Testing: Regular sampling for total viable count and endotoxins, especially from the ultrapure loop.
-
Preventive Maintenance & Sanitization: Scheduled replacement of consumables and periodic hot water or chemical sanitization.
-
Comprehensive Data Logging: Maintaining complete records of all maintenance, alerts, and water quality data for audit readiness.
L'avis d'un expert : The most common point of failure is not the purification cartridges themselves, but poor sanitization practices in the storage tank and distribution loop, leading to biofilm formation and sporadic spikes in bacteria and TOC.
Les L'eau taupe Advantage: Your Partner in Scientific Integrity
Chez Molewater, nous comprenons que votre medical water purification system is the bedrock of your lab’s operations. We provide more than equipment; we deliver guaranteed purity.
-
Application-Specific Design: Notre medical water systems are configured for your specific needs, from a clinical lab to a research core facility.
-
Validation-First Approach: We provide full IQ/OQ/PQ documentation and support to streamline your accreditation process.
-
Proactive Remote Monitoring: Our systems can offer real-time alerts and remote diagnostics to prevent unexpected downtime.
Is Your Current Water Quality Compromising Your Work?
-
Are you experiencing unexplained background noise in your assays?
-
Are your cell cultures showing inconsistent viability?
-
Is an upcoming CAP, CLIA, or FDA audit causing concern?
Stop Guessing About Your Water Quality.
Claim your FREE Laboratory Water Quality Assessment. Our specialists will review your current setup and purity requirements to identify any compliance or performance gaps.







